The formula “hcooch ch2 h2o” might look complex at first, but this chemical combination is a vital part of organic reactions, industrial production, and green chemistry. The formula captures a snapshot of an organic reaction that connects to industries from plastics and fuels to pharmaceuticals.
The combinatorial chemistry of hcooch ch2 h2o and its components—HCOOH (formic acid), CH2 (methylene), and H2O (water)—matters beyond academic circles. The reaction with the most impact in this chemical system is the hydrolysis of methyl formate. This can be represented by the balanced equation: HCOOCH₃ + H₂O → HCOOH + CH₃OH. Water acts as the nucleophile in this reaction and shows key principles of nucleophilic substitution.
Methyl formate (HCOOCH₃) has unique properties. This colorless, volatile liquid gives off a fruity smell and boils at around 32°C. You’ll find it commonly in perfumes and fragrances. The hydrolysis creates formic acid, another colorless liquid that differs with its pungent odor and a boiling point of 100.8°C. The reaction between these compounds happens mildly and cleanly—a perfect example of green chemistry in action.
Understanding the Components of HCOOCH CH2 H2O
Image Source: Morey Editions
The molecular formula HCOOCH CH2 H2O shows a chemical system with three distinct entities that work together in important ways. These components bring unique chemical properties that create versatile reaction pathways used in many industries.
Methyl Formate (HCOOCH₃) as an Ester Intermediate
Methyl formate ranks among the simplest esters that form when methanol reacts with formic acid. This colorless, volatile liquid gives off a distinctive fruity smell and has a remarkably low boiling point of 31.5°C. Its simple structure masks its chemical versatility in both industrial and laboratory settings.
Scientists have discovered that methyl formate works great as an environmentally friendly C1 building block to synthesize various carboxylic esters. It also acts as a precursor of methoxycarbonyl radical in one-carbon elongation reactions, which proves valuable in pharmaceutical synthesis. The ester has no carbon-carbon bonds, making it isomeric with acetic acid but shows different reactivity patterns.
This compound’s value extends to energy applications. As an electrochemical fuel, it delivers a favorable charge density of 12.5 MC dm⁻³, which comes close to methanol’s 14.3 MC dm⁻³ and beats formic acid’s 5.11 MC dm⁻³. The compound works well with certain metals and doesn’t damage aluminum alloys like 1050A, 6062, and 5754, even if water traces exist.
CH₂ as a Reactive Methylene Group
The methylene group (CH₂) serves as a fundamental structural motif in organic chemistry. A carbon atom bound to two hydrogen atoms connects to the rest of a molecule through two single bonds. This structure is different from methylidene groups (=CH₂), where carbon creates a double bond with the rest of the molecule.
Methylene’s reactivity comes from its electron configuration and position within molecules. Scientists call it an “active methylene” when it sits between two electron-withdrawing groups, especially carbonyl groups. The hydrogens in these active methylene groups become more acidic, which leads them to lose protons and join many organic reactions.
Methylene groups show different reactivity based on their molecular surroundings. The central methylene carbon in 1,3-dicarbonyl compounds becomes highly acidic and readily loses a proton. These properties make such compounds valuable starting materials for organic synthesis, including reactions that build carbon-carbon bonds.
Water (H₂O) as a Hydrolysis Agent
Water takes center stage in many organic transformations, especially hydrolysis reactions. In HCOOCH CH2 H2O chemistry, water serves as both solvent and reactant that helps break down larger molecules into smaller parts.
Ester hydrolysis showcases water’s nucleophilic abilities perfectly. Water molecules attack the ester’s carbonyl carbon during this process and create carboxylic acids and alcohols. Methyl formate hydrolysis specifically produces formic acid and methanol.
Reaction conditions determine water’s effectiveness in hydrolysis. Pure water alone breaks down esters slowly, but acids or bases speed up the process substantially. Acidic conditions make the carbonyl carbon more vulnerable to water molecule attacks by protonating the carbonyl oxygen. Basic conditions let hydroxide ions from water attack the carbonyl carbon directly in a process called saponification.
Acidic and basic hydrolysis show distinct reaction dynamics. Acid catalysts keep the hydrolysis reaction reversible and create an equilibrium. Basic hydrolysis, however, moves forward without reversing as the strong base becomes a reactant instead of just a catalyst. This distinction matters greatly in industrial applications where complete conversion becomes necessary.
Hydrolysis Reaction: HCOOCH₃ + H₂O → HCOOH + CH₃OH
Image Source: Digital Marketing and Digital Transformation Agency
The hydrolysis of methyl formate is a key organic transformation where water breaks an ester bond. Scientists and industries use this reaction as a model to understand ester chemistry better.
Balanced Equation and Product Formation
The hydrolysis of methyl formate follows a clear stoichiometry shown in this balanced equation:
HCOOCH₃ + H₂O → HCOOH + CH₃OH
One mole of methyl formate (HCOOCH₃) combines with one mole of water (H₂O) to create one mole each of formic acid (HCOOH) and methanol (CH₃OH). The reaction keeps perfect atom balance—every carbon, hydrogen, and oxygen atom from the reactants shows up in the products, just arranged differently.
Pure water catalyzes this hydrolysis too slowly to be useful without extra catalysts. The process needs either acidic or basic conditions to move at useful speeds. Under acidic conditions, the reaction follows this rate expression:
rate = k[HCOOCH₃][H⁺]
This rate law shows that both methyl formate and acid concentrations affect reaction speed. The H⁺ doesn’t appear in the balanced equation because it works as a catalyst.
Industrial formic acid production runs at temperatures between 90-140°C and pressures from 5-18 atmospheres. The methyl formate-water molar ratio stays between 1.5:1 and 10:1. Direct hydrolysis can get pricey because it needs extra water to work well, which makes separating products harder. Some manufacturers take a different route through formamide:
HCOOCH₃ + NH₃ → HCONH₂ + CH₃OH HCONH₂ + H₂O + ½H₂SO₄ → HCOOH + ½(NH₄)₂SO₄
Role of Water in Breaking Ester Bonds
Water’s behavior in ester hydrolysis changes based on reaction conditions. Water attacks the ester’s carbonyl carbon as a nucleophile to start breaking bonds. The mechanism changes between acidic and basic conditions.
Acidic hydrolysis starts when an acid catalyst adds a proton to methyl formate’s carbonyl oxygen, making it more electrophilic. A water molecule then attacks the carbonyl carbon to form a tetrahedral intermediate that breaks down into formic acid and methanol. This reverses Fischer esterification, making the reaction go both ways and needing extra water to push toward products.
Basic hydrolysis (saponification) works differently. Hydroxide ions attack the carbonyl carbon directly. The tetrahedral intermediate releases methoxide ions that quickly grab protons from water to make methanol. Unlike acid hydrolysis, base-catalyzed reactions only go one way because the carboxylate anion produced is less reactive than the original ester. The saponification follows this path:
HCOOCH₃ + NaOH → HCOONa + CH₃OH
Converting sodium formate to formic acid needs more acid:
HCOONa + HCl → HCOOH + NaCl
Advanced computational studies show two paths for gas-phase hydroxide-mediated hydrolysis. Hydroxide forms hydrogen bonds with the methyl group 85% of the time, while proton removal from formyl hydrogen happens 15% of the time. These theoretical calculations match what scientists see in experiments.
Manufacturers design special reactors to handle acid hydrolysis’s equilibrium challenges. Taking out reaction products (methanol or formic acid) pushes the reaction forward. Some industrial processes use a flashing step that vaporizes at least 25% of unused methyl formate, followed by vacuum distillation to separate methanol from formic acid.
Methyl formate hydrolysis is one of several ways to make formic acid, along with formamide hydrolysis and biomass oxidation. This simple-looking reaction needs complex engineering to make it profitable in industry.
Reaction Mechanism Under Acidic and Basic Conditions
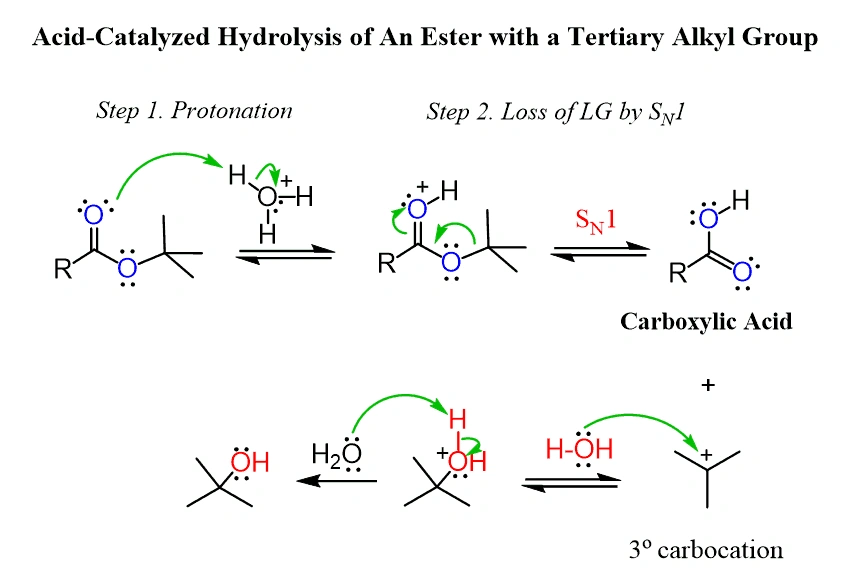
Image Source: Chemistry Steps
The reaction pathway of methyl formate hydrolysis shows different mechanisms based on pH conditions. These mechanisms show simple principles of nucleophilic acyl substitution. They also demonstrate how reaction conditions affect both kinetics and thermodynamics of organic transformations.
Protonation of Carbonyl Oxygen in Acidic Medium
Hydrolysis in acidic conditions starts when the carbonyl oxygen of methyl formate gets protonated. This creates a more electrophilic carbon center. This vital first step changes a relatively unreactive ester into a highly susceptible electrophile. The added proton increases the partial positive charge on the carbonyl carbon. It disrupts the carbonyl’s resonance stabilization.
Isotope effect studies at 20°C in 0.5 M HCl gave an explanation of this mechanism. The measured isotope effects include carbonyl carbon (¹³k = 1.028 ± 0.001), carbonyl oxygen (¹⁸k = 0.9945 ± 0.0009), nucleophile oxygen (¹⁸k = 0.995 ± 0.001), and formyl hydrogen (ᴰk = 0.81 ± 0.02). These values experimentally validate the protonation mechanism and its subsequent steps.
Nucleophilic Attack by Water Molecule
After protonation, a water molecule attacks the activated carbonyl carbon as a nucleophile. This attack happens through the Bürgi-Dunitz trajectory. The angle between the line of nucleophilic attack and the C-O bond is more than 90°. This maximizes orbital overlap between the HOMO of water and the π* LUMO of the C-O double bond.
Water alone makes the nucleophilic addition relatively slow. This explains why acid catalysis is needed for practical reaction rates. The rate of hydrolysis compared to the exchange of the carbonyl oxygen with water (kₕ/kₑₓ) is about 11.3. Under standard hydrolysis conditions, the rate of oxygen exchange measures 0.0723 min⁻¹.
Formation and Breakdown of Tetrahedral Intermediate
A tetrahedral intermediate forms from the nucleophilic attack. This structure is vital as the originally trigonal carbonyl carbon takes on tetrahedral geometry. The intermediate contains a positively charged oxonium ion that goes through several proton transfer steps:
- Deprotonation of the attacking water oxygen
- Protonation of the methoxy group to create a better leaving group
- Collapse of the intermediate with methanol departure
- Deprotonation of the resulting carboxylic acid
The tetrahedral intermediate’s stability directly affects reaction rates. Enzymes like chymotrypsin and subtilisin catalyze similar reactions. They do this by stabilizing tetrahedral intermediates. The effective concentration factors range from 41,200 M to 229,000 M compared to uncatalyzed reactions.
Saponification Pathway in Basic Conditions
The mechanism changes in basic conditions. A hydroxide ion attacks the carbonyl carbon directly instead of protonation. This skips the need for carbonyl activation. The tetrahedral intermediate that forms has a negatively charged oxygen. It then releases methoxide (CH₃O⁻) as the leaving group.
Acid and basic hydrolysis differ in their reversibility. The acid-catalyzed pathway creates an equilibrium. This makes complete conversion difficult without excess water. Basic hydrolysis (saponification) moves forward irreversibly. The carboxylate anion produced is nowhere near as electrophilic as the original ester.
The reverse reaction under basic conditions would need to form a highly unstable dianion intermediate (RC(OR)(O²⁻)²⁻). This thermodynamic barrier shows why saponification works so well to completion. Acid-catalyzed hydrolysis needs careful control of reaction conditions to push equilibrium toward products.
Physical and Chemical Properties of Reactants and Products
The HCOOCH₃ + H₂O reaction’s kinetics and handling needs depend on the physical and chemical properties of its reactants and products. These compounds have unique traits that shape their use in labs and industry.
Boiling Points and Solubility of Methyl Formate and Methanol
Methyl formate has a low boiling point of 32°C, which makes it volatile at normal conditions. Methanol boils at a higher temperature of 64.5°C, and this difference affects how industries separate these compounds. Formic acid boils at about 100°C. These stepped boiling points make fractional distillation easier during purification.
The solubility of methyl formate in water reaches 30 g/100 mL at 20°C and rises as temperature increases. This partial mixing with water affects how fast hydrolysis happens in industrial reactors. Methyl formate’s polarity parameter (ET) is 41.9 kcal/mol, which determines how it mixes with polar solvents. Its relative permittivity of 8.50 puts it between non-polar organic solvents and highly polar water.
Hydrogen Bonding in Formic Acid and Water
Hydrogen bonding shapes how formic acid and water interact during hydrolysis. Formic acid can both give and receive hydrogen bonds, unlike simple carboxylic acids. This creates a complex network of bonds, letting formic acid form multiple hydrogen bonds at once.
Research on solid argon matrices shows that water forms stable complexes with cis-formic acid at low temperatures. Strong O-H···O hydrogen bonds create this stability by stopping proton tunneling reactions. Scientists have found three main hydrogen-bonded structures between formic acid and water:
- FA-(water)₂: formic acid with two water molecules
- FA-water: simple 1:1 complex
- (FA)₂-water: two formic acid molecules with one water
Tests with vibrational spectroscopy prove that formic acid changes water’s hydrogen bond network. This affects the reaction’s mechanism and speed.
Volatility and Flammability Considerations
Methyl formate’s physical properties raise important safety concerns. It has a flash point of -19°C and can explode in air at concentrations from 4.5% to 23%. Its vapor pressure is 64 kPa at 20°C, which is much higher than methanol’s 13.0 kPa. This explains why methyl formate evaporates faster at room temperature.
The vapor from methyl formate weighs 2.07 times more than air, so it sinks and might collect in low spots. While it won’t ignite on its own until 449°C, its low boiling point (32°C) means it creates flammable vapors during normal handling.
Good ventilation is crucial when using methyl formate. People can’t smell it until it reaches about 600 ppm, but the safety limit (TLV) is just 100 ppm. This makes it dangerous to rely on smell to detect unsafe levels.
Industrial and Laboratory Applications
Image Source: Formic Acid
Products from methyl formate hydrolysis have found their way into many industries. This creates a circular economy where manufacturers and labs can use both reactants and products.
Formic Acid in Leather and Textile Processing
Formic acid is the life-blood of textile and leather industries. Textile manufacturers use formic acid-water blends to fix dyes onto fibers. The acid works as a pH regulator that will give a proper dye grip, which creates vibrant colors that last longer. Formic acid’s usefulness goes beyond just dyeing in textile production.
The leather tanning industry now depends heavily on formic acid. The global market reached USD 2.1 billion in 2023 and experts predict growth to USD 3.8 billion by 2032, with a 6.5% compound annual growth rate. We moved toward environmentally responsible leather tanning methods instead of traditional chromium-based processes. Tanners use formic acid to de-lime hides so tanning agents can penetrate better.
Methanol in Biodiesel and Fuel Cells
Methanol, which comes from methyl formate hydrolysis, plays a crucial role in making biodiesel. Manufacturers can produce industrial-scale biodiesel with 98% purity through transesterification of triolein and methanol using catalysts like sodium hydroxide or sulfuric acid. Modern methanol synthesis uses reforming technologies on coke oven gas and carbon dioxide or glycerol—a biodiesel synthesis byproduct.
Methanol also serves as an energy carrier in next-generation fuel cells. Life Cycle Assessment shows that making methanol from coke oven gas and carbon dioxide, then producing biodiesel through alkali catalysis offers the most environmentally friendly option.
CH₂ in Polymer and Resin Synthesis
The methylene (CH₂) group aids various polymer synthesis applications. Resins with customizable mechanical properties emerge when di- or tri-functional CH₂-bearing monomers react under reflux conditions. Methylene derivatives work best to adjust polymer cross-linking density, which boosts rubber products’ elasticity and mechanical strength.
Polystyrene resins with phosphonium chloride or bromide effectively catalyze complex syntheses like [MoCl(CO)2(η3-CH2C(COCl)C = CH2)L2]. The polymer structure controls conversion rates. Adding amines or amino acids creates amide 2-substituted η3-butadienyl complexes.
Use in Catalyst Screening and pH-Controlled Reactions
Labs use the HCOOCH CH2 H2O system to screen catalysts. They test hydrogenation or radical coupling reactions with trace metal salts like Pd²⁺ and Cu²⁺. GC-MS monitoring techniques track how these reactions progress.
pH-controlled reactions need careful formic acid titration into aqueous systems. Scientists must keep acidity levels between pH 2-4 for condensation reactions. This precise pH control leads to reproducible results, especially when you have hydrolysis reactions where acidity levels directly influence product yields and purity.
Safety, Handling, and Environmental Impact
Working with HCOOCH CH2 H2O components needs strict safety protocols because each substance has its own hazards.
Toxicity and Flammability of Methyl Formate and Methanol
Methyl formate needs careful handling because it is very flammable with a flash point of -2°F and explosive limits ranging from 4.5% to 23% in air. It can cause respiratory irritation, central nervous system depression, and visual disturbances when inhaled. Methanol’s toxicity concerns are even greater – a dose of approximately 1 g/kg body weight could be lethal. People who get methanol poisoning don’t show severe symptoms at first. A latent period of 6-30 hours follows before metabolic acidosis develops. Methanol’s toxic metabolite, formic acid, causes permanent visual deficits and can lead to coma or death.
Corrosive Nature of Formic Acid
Formic acid’s corrosive properties can cause severe burns to any body part it touches. Skin contact leads to pain, blistering, and ulceration, while eye contact results in pain, inflammation, and possible permanent damage. Industrial settings must use acid-resistant materials to prevent equipment degradation.
Effluent Treatment and Green Chemistry Benefits
The wastewater from these reactions is relatively harmless and contains mostly biodegradable methanol and formic acid. The hydrolysis reaction creates no wastewater discharge, waste gas, or residue, which lines up with green chemistry principles. Both formic acid and methanol break down naturally, so their effect on the environment is minimal when handled properly.
Conclusion
The hydrolysis of methyl formate is a perfect example of organic chemistry that connects simple principles with real-world uses. This reaction turns methyl formate and water into formic acid and methanol. The process takes two different paths under acidic and basic conditions. Acidic hydrolysis works through carbonyl activation and nucleophilic attack. It creates an equilibrium that chemists must carefully control to complete the reaction. Basic hydrolysis takes a different route through irreversible saponification, which makes it better to get complete conversion.
This simple chemical system powers many industrial processes. The formic acid we get from this reaction is a great way to get better results in leather tanning and textile dyeing. It creates bright, lasting colors and offers better environmental options than chromium-based methods. The other product, methanol, plays a key role in making biodiesel and next-generation fuel cells, which helps create sustainable energy solutions.
Safety must come first when handling these compounds. Methyl formate’s low flash point and high volatility make it highly flammable. Formic acid’s corrosive nature can cause severe burns if it touches skin. Methanol might be the riskiest of all. Its toxicity tricks you – mild symptoms show up first, but they can lead to dangerous metabolic acidosis.
In spite of that, this chemical system fits well with green chemistry principles. Formic acid and methanol break down naturally, which leaves little environmental impact when handled properly. The reaction produces no wastewater or leftover residue, making it environmentally friendly under the right conditions.
The HCOOCH CH2 H2O system goes beyond just classroom learning. It gives chemists and engineers powerful tools to create valuable products while protecting the environment. This reaction system’s flexibility, efficiency, and minimal waste will keep it relevant in many industries, from pharmaceuticals to textiles, as sustainable chemistry becomes more crucial.
FAQs
1. What is the main reaction involved in HCOOCH CH2 H2O chemistry?
The main reaction is the hydrolysis of methyl formate, represented by the equation HCOOCH₃ + H₂O → HCOOH + CH₃OH. This reaction produces formic acid and methanol from methyl formate and water.
2. How does the reaction mechanism differ in acidic and basic conditions?
In acidic conditions, the carbonyl oxygen is protonated first, followed by nucleophilic attack from water. In basic conditions, hydroxide ions directly attack the carbonyl carbon. Acidic hydrolysis is reversible, while basic hydrolysis (saponification) is irreversible.
3. What are some industrial applications of the products from this reaction?
Formic acid is used in leather tanning and textile dyeing. Methanol is important in biodiesel production and fuel cells. The methylene group (CH₂) is used in polymer and resin synthesis.
4. What safety precautions should be taken when handling these chemicals?
Methyl formate is highly flammable and volatile. Formic acid is corrosive and can cause severe burns. Methanol is toxic if ingested. Proper ventilation, protective equipment, and careful handling procedures are essential when working with these substances.
5. How environmentally friendly is this chemical system?
The hydrolysis reaction produces no wastewater discharge, waste gas, or residue. Both formic acid and methanol are biodegradable, making their environmental impact minimal when properly managed. This aligns well with green chemistry principles.

